You are using an unsupported browser. Please use the latest version of
Chrome, Firefox, Safari or Edge.
Stories
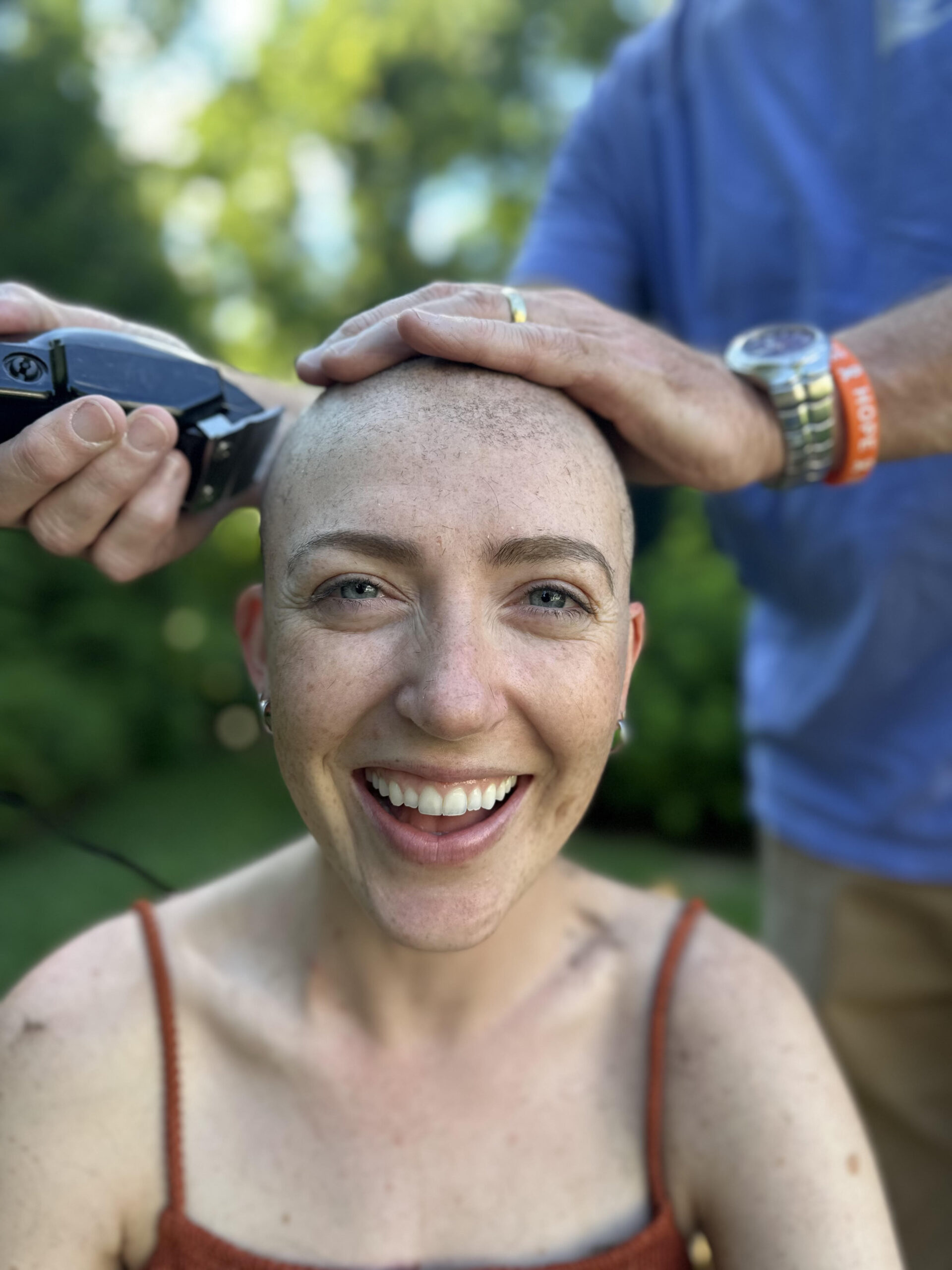
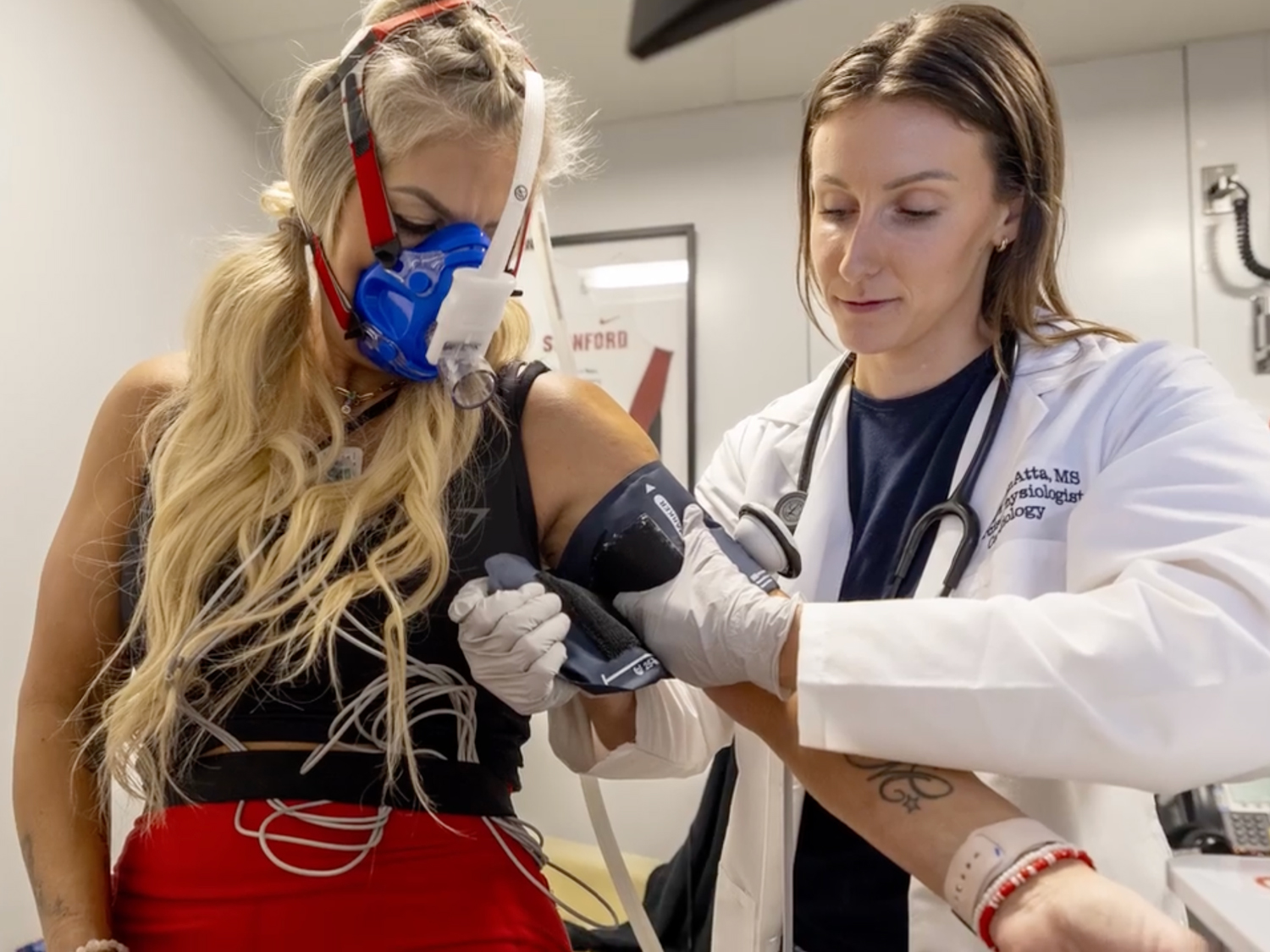
Inspiration. You can find it in every Mass General story. In every hand we hold, and every community partnership and global relief effort we launch. In the face of our newest medical resident and in our latest research breakthrough, decades in the making.
These stories capture how Mass General is harnessing inspiration to make a difference in the lives of patients, families, and our local and global neighbors. Read on. Get inspired. Join us.