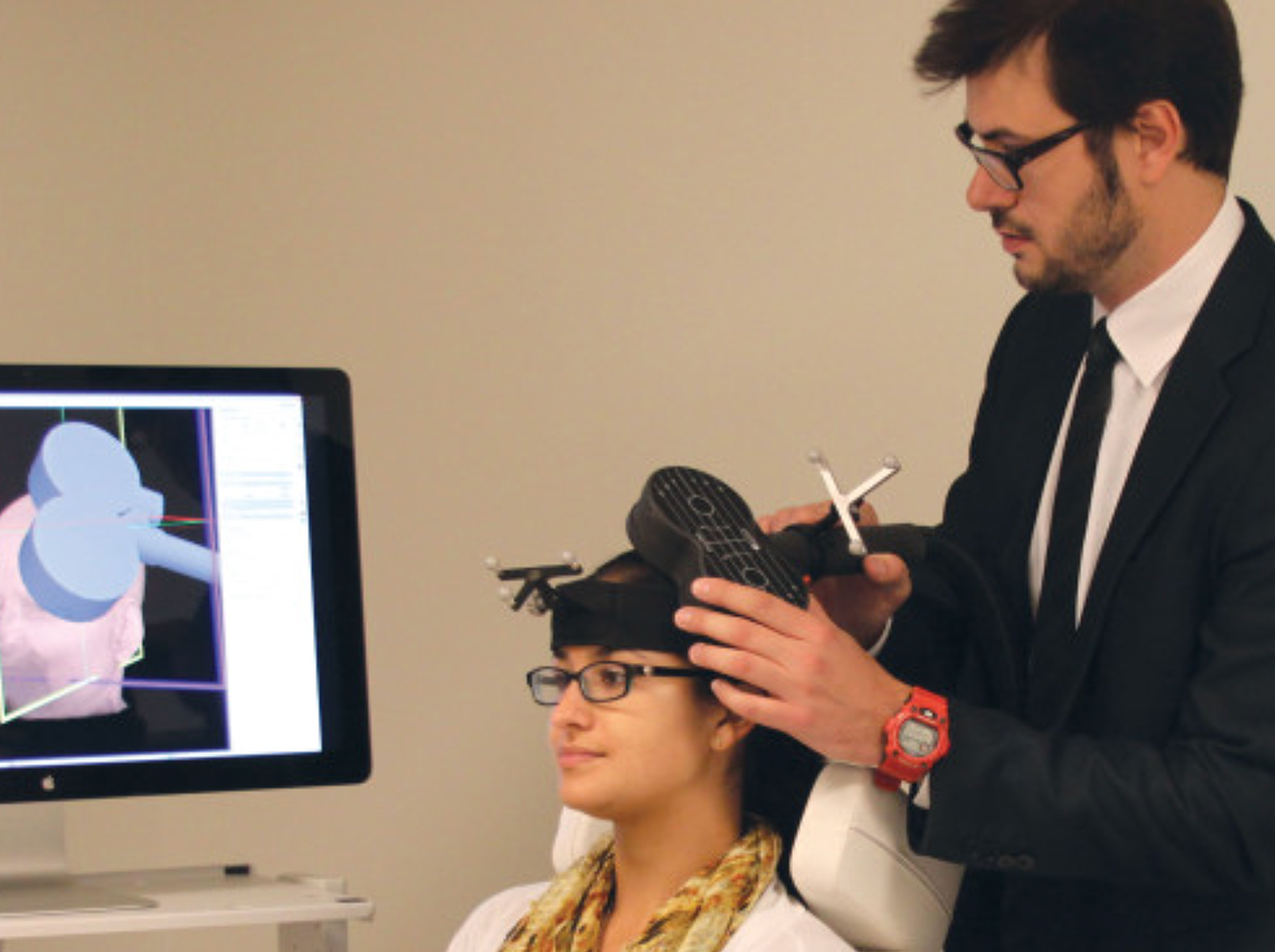
Pictured above, Joan Camprodon, MD, PhD, MPH, demonstrates the use of TMS on research assistant, Amanda Arulpragasam.
For patients suffering from depression, the Massachusetts General Hospital Department of Psychiatry now offers another treatment option — a new clinic based in Charlestown that uses transcranial magnetic stimulation (TMS). The clinic is part of a broader effort that includes a research enterprise program, both led by Joan Camprodon, MD, PhD, MPH, director of the Laboratory for Neuropsychiatry and Neuromodulation at Mass General. Dr. Camprodon’s team aims to understand how the brain’s structure and function affect disease and how interventions such as TMS can change the mechanisms that contribute to disease.
During a TMS procedure, focused magnetic impulses are directed to the brain. The electrical currents produced stimulate nerve cells involved in mood regulation that may be underactive in diseases such as depression. By restoring the equilibrium, TMS helps reset this imbalance of chemicals. from a diseased brain to a healthy one.
TMS: Safe and Effective
Approved by the U.S. Food and Drug Administration for treating depression, TMS is safe, noninvasive and effective. “It’s a good alternative for people who can’t or don’t want to take drugs,” Dr. Camprodon says. “We know that it works, and patients don’t have to endure side effects from medication.”
Another benefit is the integration and co-location of the research program and the clinical service. When scientists develop new techniques or discover new knowledge, this information can immediately be put into practice to benefit patients. Initially, the program’s focus is on adults, but the plan is to make this treatment safe for children and adolescents in the future. “The results can be life changing,” Dr. Camprodon says. “We can improve function and well-being without adding another drug.”
To learn more about the Department of Psychiatry at Mass General, please contact us.