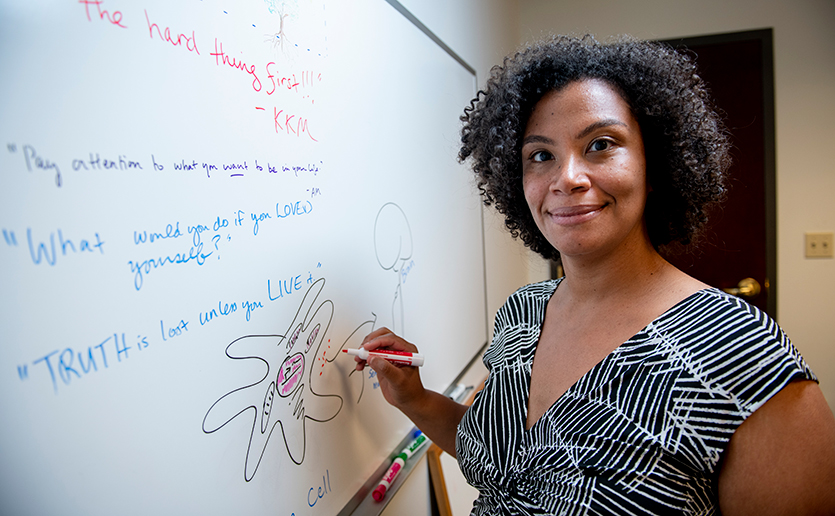You are using an unsupported browser. Please use the latest version of
Chrome, Firefox, Safari or Edge.
Stories
Inspiration. You can find it in every Mass General story. In every hand we hold, and every community partnership and global relief effort we launch. In the face of our newest medical resident and in our latest research breakthrough, decades in the making.
These stories capture how Mass General is harnessing inspiration to make a difference in the lives of patients, families, and our local and global neighbors. Read on. Get inspired. Join us.
Innovation Story
Simmie L. Foster, MD, PhD, Explains Hot and Cool Research
Innovation Story
The Wild West of “Medicinal” Cannabis
Innovation Story
Improving Outcomes for Bipolar Disorder
Profile in Medicine
Using Life’s Experiences to Determine the Meaning of Home
Profile in Medicine
Welcomed, With Open Arms
Innovation Story
Five Things Mass General is Doing to Improve Sustainability
Donor Story
Brave Theo
Patient Story
The Heart of an Athlete
Innovation Story
Loneliness: An Unmet Need for Social Connection
Innovation Story